Designing a medical product isn’t just about innovation and usability, it’s also about meeting strict regulatory requirements. The U.S. Food and Drug Administration (FDA) sets clear expectations for medical devices to ensure patient safety and product effectiveness. For innovators, this means FDA compliance must be built into the design process from the start.
At CHOI Design, we’ve helped innovators bring FDA-compliant products to life—like intuitive Clarix X-Ray systems for quick, 3D tissue imaging and sleek diagnostic tools that prioritize usability and patient safety. These real-world projects reflect our deep expertise in navigating medical device design from concept through compliance.

This guide explores how product designers can navigate FDA regulations at a high level—from usability strategies to device classification—while revealing how a collaborative, strategic design partner can accelerate compliance and market success.
Why FDA Compliance Matters in Medical Product Design
Medical devices directly impact human health, making them highly regulated. The FDA ensures each device is safe, reliable, and effective (FDA Overview of Device Regulation).
Delaying compliance planning often leads to:
- Costly redesigns late in development
- Launch delays and approval hiccups
- Increased liability exposure
- Damaged brand reputation
By integrating FDA requirements from day one, teams reduce risk and boost confidence in outcome.
Understanding FDA Device Classification
FDA divides devices into three risk classes:
- Class I – Low risk (e.g., bandages); generally only basic quality controls are needed.
- Class II – Moderate risk (e.g., infusion pumps); require additional safeguards and “substantial equivalence” reviews.
- Class III – High risk (e.g., pacemakers); require rigorous clinical evidence and approval.
Most innovations fall into Class I or II, where thoughtful design and strong documentation can be decisive.
The Role of Human Factors in Design
A device might work perfectly—but if it’s not intuitive, it still poses risk. That’s why the FDA emphasizes human factors engineering.
Best practices include:
- Simple, intuitive interfaces to reduce errors
- Designing for diverse users—patients, clinicians, caregivers
- Early prototype testing to uncover usability challenges
- Clear documentation of findings and revisions
At CHOI Design, our prototyping and user testing process empowers teams to catch design flaws early and align with FDA expectations.
Embedding Quality into the Design Process
The FDA’s Quality and Compliance for Medical Devices guidance emphasizes that quality must be integrated into every step—not tacked on later.
For design teams, this translates into:
- Early risk assessments and iterative prototyping
- Clear, version-controlled documentation of design decisions
- Cross-functional collaboration across design, engineering, and regulatory teams
This disciplined approach doesn’t just satisfy regulators; it ensures product quality across its lifecycle.
Prototyping: A Strategic Advantage for Compliance
Prototypes are more than design mockups—they are compliance deliverables. They allow teams to:
- Validate design against regulatory expectations
- Test materials for safety and function
- Capture real-world user feedback
- Fine-tune manufacturability
Every stage of prototyping builds stronger evidence for FDA review.
Simplifying FDA Documentation
FDA expects clear records of design, testing, and validation processes (FDA Products and Medical Procedures). It may sound daunting, but the principle is simple: traceability matters.
Focus on capturing:
- How user needs influenced design
- Updated risk assessments
- Prototype testing results and revisions
- Versioned design outputs for accountability
Strong documentation doesn’t just facilitate approval—it future-proofs your product for iterative improvements.
Why a Collaborative Approach Wins with CHOI Design
FDA compliance isn’t a solo journey—it’s a team sport. When designers, engineers, and regulatory experts work in sync, compliance happens naturally.
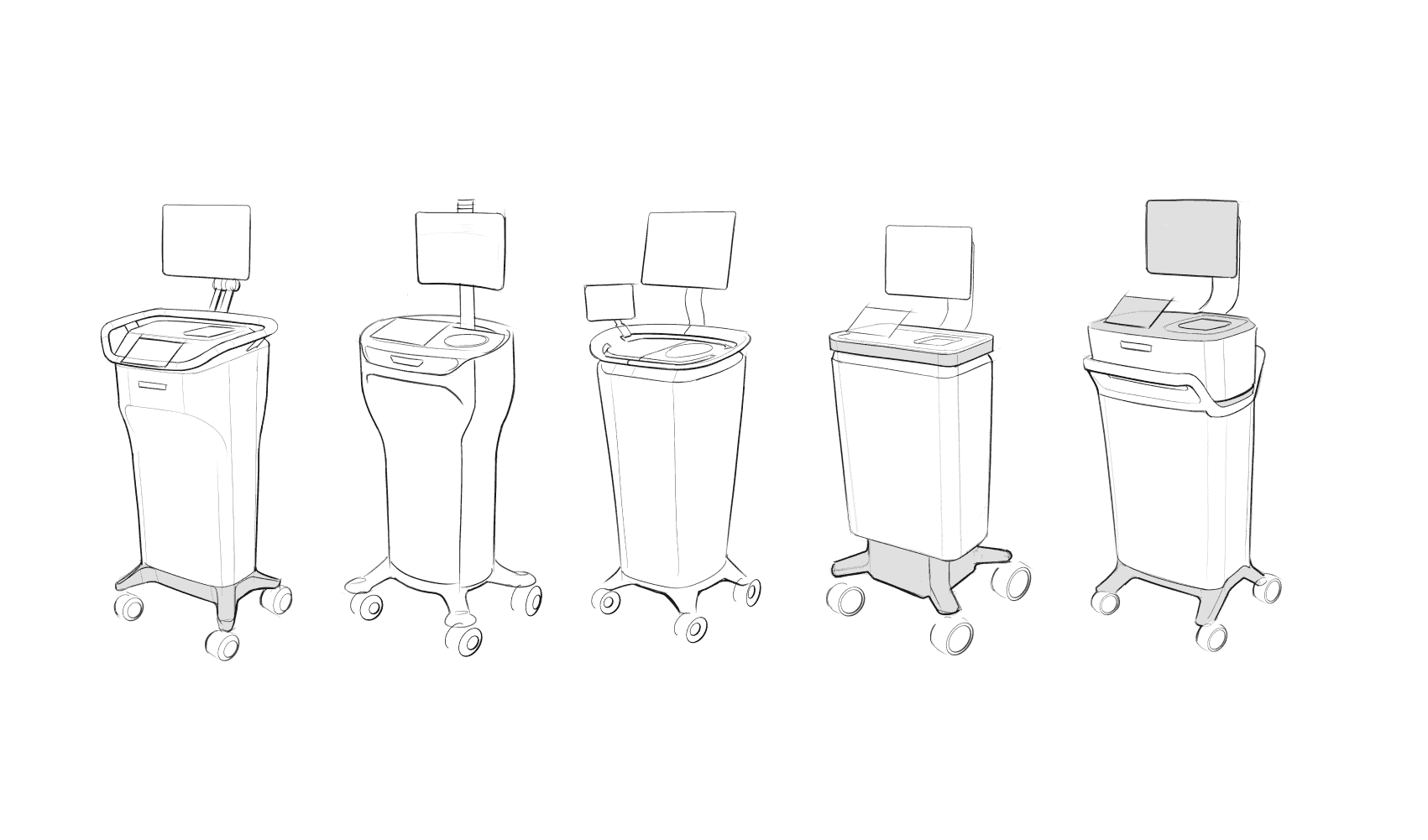
CHOI Design brings decades of experience delivering compliant medical systems—from Class I diagnostic tools to Class II imaging technology like our Clarix X‑Ray solutions. As strategic partners, we help our clients:
- Reduce risk and ambiguity
- Accelerate development timelines
- Deliver products that meet FDA standards and user expectations
Let’s Bring Your Medical Innovation To Market
FDA compliance isn’t a hurdle—it’s a chance to craft safer, smarter, and more trusted products. Thoughtful design makes the difference between a product that merely meets standards and one that truly excels in the market.
CHOI Design blends design insight, regulatory awareness, and real-world experience to deliver compliant medical products that resonate with users and perform under scrutiny. Whether you’re designing low-risk or advanced systems, our team supports you through every step—from strategy to launch.
Ready to bring your medical innovation to market with confidence? Contact us to explore how we can partner in your journey.